Welcome to BAR - the Bay Area's premier saltwater hobbyists hub!
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
"The Lab" - Josh and Tiffany's IM Nuvo EXT 200
- Thread starter IOnceWasLegend
- Start date
that is correct, i had 10x xr15pros g5 over a 48x36 tank. at one point i had 12 xr15s just for fun.I'm slowly going to raise up the PAR, since I'm currently running these lights at only about 65%. I just don't want to bake my torches just yet.
EDIT: IIRC, he had XR15s (not XR30s) and G5s vs G6. I'd argue that 10x XR15s outpeform 4x XR30s, but the light spread is noticeably better on the G6s so it's probably not *too* bad once I bump it up a bit more after getting more acros.
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #223
4-29-24
Photo dump.







Photo dump.
Supporting Member
- Joined
- Dec 16, 2020
- Messages
- 1,354
i recommend 2 led bars, great coverage, helps with shadowing of sps and coral (later on), and par increase also nice coral pop. Good one is OR3 Blue bar from orphrek or reef bright. at price of one xr15 you get two OR3 Blue light bars (48inch bar $200/ 60inch $325)
- Joined
- May 24, 2022
- Messages
- 5,252
Quantas are a pretty good option too. 6' bar is like $300i recommend 2 led bars, great coverage, helps with shadowing of sps and coral (later on), and par increase also nice coral pop. Good one is OR3 Blue bar from orphrek or reef bright. at price of one xr15 you get two OR3 Blue light bars (48inch bar $200/ 60inch $325)
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #226
5-6-2024
Ignoring the testing reagent change + subsequent calibration...that's some mighty fine alk stability.
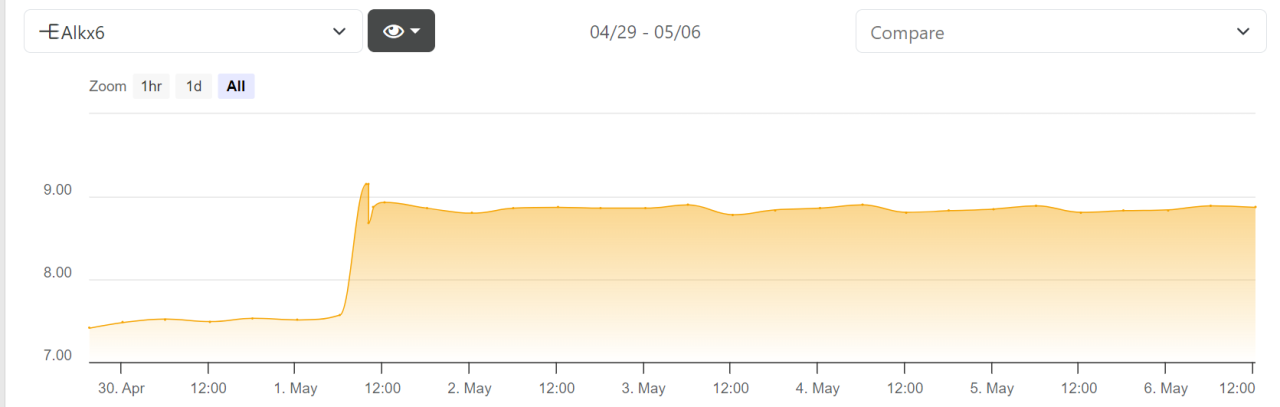
Ignoring the testing reagent change + subsequent calibration...that's some mighty fine alk stability.
- Joined
- Aug 19, 2023
- Messages
- 4,559
Noob question here, is that chart from a trident system or something similar?5-6-2024
Ignoring the testing reagent change + subsequent calibration...that's some mighty fine alk stability.
View attachment 55737
I'm assuming is not from any of the diy test kits since it's showing daily values.
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #228
It's from the Trident. It automatically logs your test results whenever it tests (in my case, 4x/day).Noob question here, is that chart from a trident system or something similar?
I'm assuming is not from any of the diy test kits since it's showing daily values.
- Joined
- Aug 19, 2023
- Messages
- 4,559
Thanks it's something that's on my future wish list lol, my wife wouldn't support that type of venture presently.It's from the Trident. It automatically logs your test results whenever it tests (in my case, 4x/day).
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #230
Keep an eye out in the coming months; if the Maven is as good as it's touted to be (from Hydros) there will likely be a lot of used Tridents on the market soon.Thanks it's something that's on my future wish list lol, my wife wouldn't support that type of venture presently.
Last edited:
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #231
Not a tank update, but my wife won me a stuffed puffer fish dressed as a clownfish!

- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #232
7-10-24
Tank's been chugging along well. No further losses of torches (which makes me more convinced it was, in fact, something that was treated by the interceptor). We've also slowly but surely been adding acros to the tank, and I'll have all the goodies I got from the swap (and a TCK master torch) ready to come out of QT next week! For now: photo dump.










Tank's been chugging along well. No further losses of torches (which makes me more convinced it was, in fact, something that was treated by the interceptor). We've also slowly but surely been adding acros to the tank, and I'll have all the goodies I got from the swap (and a TCK master torch) ready to come out of QT next week! For now: photo dump.
Supporting Member
- Joined
- Mar 19, 2024
- Messages
- 2,549
omgg!!Not a tank update, but my wife won me a stuffed puffer fish dressed as a clownfish!
View attachment 55906
- Joined
- May 24, 2022
- Messages
- 5,252
7-10-24
Tank's been chugging along well. No further losses of torches (which makes me more convinced it was, in fact, something that was treated by the interceptor). We've also slowly but surely been adding acros to the tank, and I'll have all the goodies I got from the swap (and a TCK master torch) ready to come out of QT next week! For now: photo dump.

Past President
- Joined
- Mar 26, 2010
- Messages
- 18,870
I know who I’m hitting up if I ever get a tank going again!
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #236
Update 7-14-24
Coral QT tank has had flatworms for a few weeks now; I've been religiously dipping the corals with potassium chloride every other day for the last week, but I'm still getting a few off every dipping. My suspicion is that these are the harmless red planaria making their way onto the coral. However, since I'm pretty sure a few coral had euphyllia eating flatworms at some point (given how irritated they were with flatworms on their flesh), I don't want to risk it. So today I bit the bullet and used Flatworm eXit. Given there were enough flatworms I could see them on the glass, I was very careful and did the following:
Next steps/plans for the tank
The big one is I'll be swapping the tank over from Neptune Apex to CoralVue Hydros. I currently have an X4 running on the QT/frag tank and have really liked it, as well as trusting the components to be a bit more robust/water-resilient than Apex. My planned setup for the display will be:
Coral QT tank has had flatworms for a few weeks now; I've been religiously dipping the corals with potassium chloride every other day for the last week, but I'm still getting a few off every dipping. My suspicion is that these are the harmless red planaria making their way onto the coral. However, since I'm pretty sure a few coral had euphyllia eating flatworms at some point (given how irritated they were with flatworms on their flesh), I don't want to risk it. So today I bit the bullet and used Flatworm eXit. Given there were enough flatworms I could see them on the glass, I was very careful and did the following:
- Removed all snails from the tank, rinsed them in fresh saltwater, and put them into my display tank (since they'd been in the tank for the full QT period)
- Removed all the coral from the tank, dipped in potassium chloride, and placed them in a bucket of fresh saltwater as a holding tank
- Treated the tank with Flatworm eXit following manufacturer's instructions
- After 30 minutes and seeing flatworms floating around the tank, performed a ~90% water change
- Ran activated carbon for 30 minutes on the tank to clear out any traces of toxin
- Put the coral back in the tank
- Monitored closely for any signs of corals being pissed off
Next steps/plans for the tank
The big one is I'll be swapping the tank over from Neptune Apex to CoralVue Hydros. I currently have an X4 running on the QT/frag tank and have really liked it, as well as trusting the components to be a bit more robust/water-resilient than Apex. My planned setup for the display will be:
- Monitoring: Hydros X4 (1x pH probe for tank, 1x pH probe for calcium reactor)
- Control: Hydros XP8 energy bar
- Testing: Hydros Maven
- Dosing: Hydros Sole
- The X10 is based on pH and acid titration (similar to KH Guardian and KH Director), while the Maven will is based on colorimetric readings (similar to the Trident). pH & acid titration is cheaper in the long run and you can DIY your reagent; however, a lot of the reports I read indicated bubbles in the dosing lines could throw off readings. While this isn't a huge deal (I think it was usually in the range of 0.3-0.4 dKH flux), minimal maintenance is absolutely a priority to me.
- Calcium testing on the Maven; since I run a calcium reactor, calcium creeps up over time and I'd like to automatically keep an eye on it, rather than testing manually.
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #237
Update 2-25-25
It’s been a while since my last update, and unfortunately not a happy one (though hopefully useful for others).
~9-10 months ago, I started having another round of torches dying off. It was identical to the last occurrence, and was a repeat of the same issue that’s plagued me for about four years now:
(You can skip ahead at this point, since I’m just doing a comprehensive rundown of what I’ve tried for those trying to troubleshoot in the future)
I figured there were several possibilities: it could be chemistry/nutrient-related. It could be a fish biting/harming the coral. It could be parasites. It could be bacterial. Going through all of these:
The only thing I could potentially think of at the time was something biological. It made sense: the issues has followed me through four separate tanks now. It waxes and wanes (consistent with shifting diversity and general system health). The coral dying was not brown jelly disease, but that didn’t necessarily rule out bacteria or something else. So I did both eukaryotic and bacteria Aquabiomics tests to try and get an answer.
While I’m generally skeptical about drawing conclusions from these tests—specifically because there’s a big gulf between “having X proportion of Y bacteria in your tank” and “that observation being the cause of the underlying dysfunction in your tank”—I did get one very interesting result.

Two things spring to mind. The first is that I have a ~15x higher incidence of this bacteria than the average tank they test (and 5x higher than the ‘high’ end of the typical range). The second is that phrase SCTLD, which I hadn’t heard of before. So I did a bit of digging.
It turns out that SCTLD stands for Stony Coral Tissue Loss Disease. The symptoms, interestingly enough, seem highly consistent with what I’ve experienced. Doing more digging, research articles have shown it can be transferred via infected coral tissue, water, and sediment (which is consistent with this issue following me through several tanks). Adding one additional piece of evidence, the only soft coral in our tank—a magic carpet mushroom—is thriving and happy. The down side, however, is that there’s no clear consensus if this issue is bacterial, viral, eukaryotic, or ‘some combination of all of the above.’
Additionally, @Thales was kind enough to discuss my issues with me after the frag swap this past weekend, and agreed that my issue sounded like it was either bacterial or confounded by bacteria. After discussing his bout with a previous, superficially similar issue, he brought up that oxolinic acid had seemingly resolved his issue (as well as admitting that, “Well, you’re basically in uncharted territory.”)
So, still no a guarantee, but: it’s a starting point.
Making a plan
After all of the above, I’m opting to try a treatment strategy with oxolinic acid with the following rationale:
Finally, I’m going to be experimenting with another veterinary-adjacent company—MiDog DNA—that claims to do marine DNA testing. I’m getting more information from them right now, but looking into what they have publicly available it seems like they run on the same ‘backbone’ that Aquabiomics does (16S ribosomal RNA sequencing). Best of all, it seems they offer a turnaround time of 2–5 days (which would let me immediately jump to another treatment if this one fails), rather than Aquabiomics’ rather…let’s say lengthy turnaround time.
It’s been a while since my last update, and unfortunately not a happy one (though hopefully useful for others).
~9-10 months ago, I started having another round of torches dying off. It was identical to the last occurrence, and was a repeat of the same issue that’s plagued me for about four years now:
- A torch colony—or just one head, or several heads on the same colony—would inflate less
- The flesh would start to recede from the bottom up
- The flesh would start to ‘peel’ off from the torch
- The flesh would slough off, and the torch would die; anywhere from several days to several weeks following the initial symptoms
(You can skip ahead at this point, since I’m just doing a comprehensive rundown of what I’ve tried for those trying to troubleshoot in the future)
I figured there were several possibilities: it could be chemistry/nutrient-related. It could be a fish biting/harming the coral. It could be parasites. It could be bacterial. Going through all of these:
- Chemistry: I test four times a day with my Trident; similarly, I test nutrients every week or two with Hanna checkers. I won’t dig through my logs for exact numbers, but nothing was consistently out of line in a way that made me think chemistry was an issue. ICP tests confirmed this, with the only consistent issue being lower-than-ideal iodine.
- Fish issues (or ‘fisshues’): One immediate possibility was our aiptasia-eating filefish (I’ve had issues with one in the past nipping at acans and fleshy corals). However, after observing it for long periods of time, I’d lean towards fish not being the cause of my issues (especially since I feed fairly heavily).
- Parasites: Everything added to my display tank has gone through a full quarantine including visual inspection and dips in CoralRx and/or potassium chloride.
- Flatworms were an initial obvious concern, but 1) all coral going into QT is visually inspected, 2) they’re dipped multiple times every few days if evidence is found, 3) the QT was treated with Flatworm eXit several times, and 4) I have a pearly wrasse in the DT for biological control.
- I initially thought euphyllia-eating white bugs may be the culprit, given I saw some on my dying reverse tiger torch. However, treating both the main display and QT tank with Interceptor had no effect on the recurrence of this issue.
- Bacterial: See below
The only thing I could potentially think of at the time was something biological. It made sense: the issues has followed me through four separate tanks now. It waxes and wanes (consistent with shifting diversity and general system health). The coral dying was not brown jelly disease, but that didn’t necessarily rule out bacteria or something else. So I did both eukaryotic and bacteria Aquabiomics tests to try and get an answer.
While I’m generally skeptical about drawing conclusions from these tests—specifically because there’s a big gulf between “having X proportion of Y bacteria in your tank” and “that observation being the cause of the underlying dysfunction in your tank”—I did get one very interesting result.
Pictured: One very interesting result
Two things spring to mind. The first is that I have a ~15x higher incidence of this bacteria than the average tank they test (and 5x higher than the ‘high’ end of the typical range). The second is that phrase SCTLD, which I hadn’t heard of before. So I did a bit of digging.
It turns out that SCTLD stands for Stony Coral Tissue Loss Disease. The symptoms, interestingly enough, seem highly consistent with what I’ve experienced. Doing more digging, research articles have shown it can be transferred via infected coral tissue, water, and sediment (which is consistent with this issue following me through several tanks). Adding one additional piece of evidence, the only soft coral in our tank—a magic carpet mushroom—is thriving and happy. The down side, however, is that there’s no clear consensus if this issue is bacterial, viral, eukaryotic, or ‘some combination of all of the above.’
Additionally, @Thales was kind enough to discuss my issues with me after the frag swap this past weekend, and agreed that my issue sounded like it was either bacterial or confounded by bacteria. After discussing his bout with a previous, superficially similar issue, he brought up that oxolinic acid had seemingly resolved his issue (as well as admitting that, “Well, you’re basically in uncharted territory.”)
So, still no a guarantee, but: it’s a starting point.
Making a plan
After all of the above, I’m opting to try a treatment strategy with oxolinic acid with the following rationale:
- My issue is likely biological, given it’s followed me from tank to tank and does not appear to be either chemistry-, fish-, or parasite-based.
- The strongest lead at the moment is an overabundance of bacteria associated with SCTLD, which is consistent with the issues I’m experiencing
- @Thales had a prior issue superficially similar to mine, which cleared up after treatment with an antibiotic (oxolinic acid)
- Oxolinic acid is relatively reef-safe and is less likely to cause any nasty side effects
Finally, I’m going to be experimenting with another veterinary-adjacent company—MiDog DNA—that claims to do marine DNA testing. I’m getting more information from them right now, but looking into what they have publicly available it seems like they run on the same ‘backbone’ that Aquabiomics does (16S ribosomal RNA sequencing). Best of all, it seems they offer a turnaround time of 2–5 days (which would let me immediately jump to another treatment if this one fails), rather than Aquabiomics’ rather…let’s say lengthy turnaround time.
Last edited:
- Joined
- Feb 5, 2020
- Messages
- 3,014
- Thread Author
- #239
Nautella is gram-negative, and oxolinic acid targets gram-negative bacteria. I'll likely try reseeding my tank in the near future, but I want to wait and see what the microbiome balance looks like after oxolonic acid treatment prior to doing so.Have you done any research on which antibiotics Nautella sp. may be particularly susceptible to? Have you thought about seeding your tank with bacteria from a healthy tank right after antibiotic treatment?
- Joined
- May 24, 2022
- Messages
- 5,252
They were using amoxicillin with denture cream to treat montastrea in the Caribbean. But that might be a hammer rather than a scalpel since it's effective against gram positive and negative. If I recall they don't have a specific type of "cause" for SCTLD.Have you done any research on which antibiotics Nautella sp. may be particularly susceptible to? Have you thought about seeding your tank with bacteria from a healthy tank right after antibiotic treatment?
About Us
Welcome to BAR!
Bay Area Reefers (BAR) is a club for reef aquarists in the San Francisco Bay Area. The primary purpose of BAR is to promote, foster, and encourage education and appreciation for the ethical husbandry and propagation of marine life.
-The Board of Directors (BOD)
